Business Operation
Our corporate mission is to provide immunology-based,
innovative new drugs for the cancer treatment to health care sites.
For this purpose, the company has been developing the drug products in the following four fields.
Our 4 Business contents
Features of Our Technology
While ImmunoFrontier, Inc has several core technologies, “CHP technology” is expected to be put into practical use ealiest of all. Wide range of application is now in progress to CHP cancer protein vaccines and next generation CHP bio-medicines. We are also trying to establish various new technologies for promoting the creation of next generation cancer DNA vaccines and therapeutics for immune diseases.
CHP Technology and Its Application to Cancer Protein Vaccines
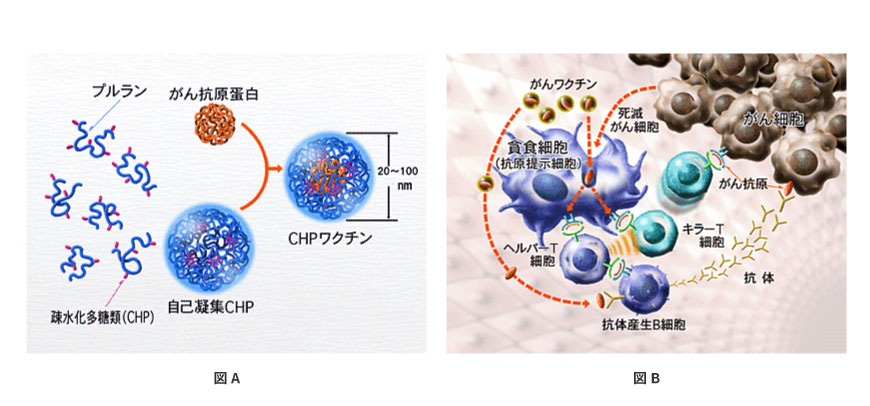
CHP is short for Cholesteryl Hydrophobized Pullulan. Pullulan, a natural polysaccharide, is chemically modified with cholesterol to add hydrophobicity. CHP forms spherical particles of nanometer size by self-organization under the physiological condition (see Fig. A). CHP particle can contain various substances in it and especially, it is a very unique material excellent in embedding proteins. While CHP has an action to help stabilization and folding of proteins by this nature, It showed a very interesting nature when CHP was applied to vaccination with protein antigens. That is the finding that when antigen proteins are immunized in the form of complex with CHP, reaction of both killer and helper T cells specific to the antigen proteins is strongly and simultaneously activated.
As it is well known in the field of vaccine that when immunization is made only with proteins alone, only helper T cells are usually activated, resulting in vaccines of lower performance, the fact that antigen proteins administered as a complex with CHP could activate both antigen-specific killer and helper T cells attracted wide attention. At the same time, it was a great discovery that made us to decide the R&D of CHP cancer protein vaccines. The mechanism that antigen protein can activate both killer and helper T cells when immunized in a form of complex with CHP is now under investigation in detail. The outline is illustrated in Fig. B.
Cancer DNA Vaccines
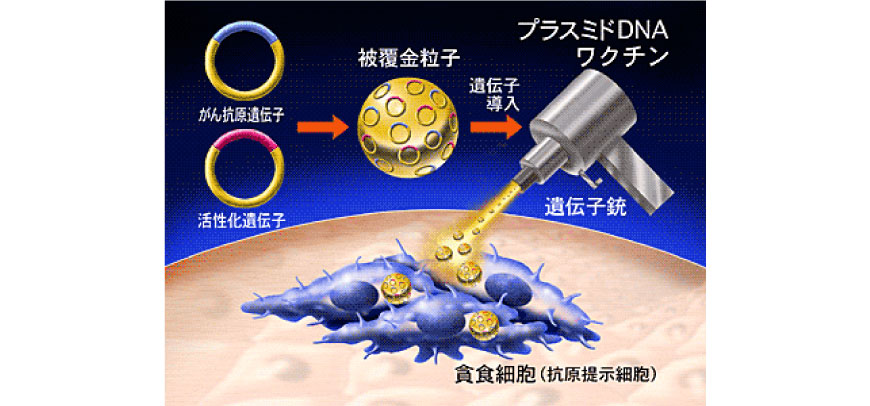
Technology of DNA vaccines is the most advanced immunization method which utilizes molecular biology techniques that inoculates antigens into the body in the form of “genes” encoding antigen proteins. DNA vaccines can be administered in a form of plasmid DNA or viral vectors, etc. For example, when DNA vaccines are subcutaneously administered as plasmid DNA, the plasmid DNA is incorporated into antigen presenting cells and produces the intended antigen protein (see the following Figure). Thereafter, like protein vaccines, antigen protein is presented to and activate antigen-specific T cells.
Compared with protein vaccines, DNA vaccines have some advantages. For example, the manufacturing process is relatively easier. In addition, it is one of the features of DNA vaccines that there is large room for further improvement of making it more efficient. Paying attention especially to this point, we are developing “multifunctional and highly efficient cancer DNA vaccines” carrying genes of various immunostimulating molecules, in addition to genes encoding antigen protein, in order to totally activate the immune system. Some prototypes of our DNA vaccines are now under evaluation. For example, we are conducting the preclinical efficacy testings of prototype DNA vaccines carrying multiple components of antigen proteins for killer and helper T cells and immunostimulating molecules that activate antigen presenting cells and T cells and signaling molecules that suppress regulatory T cells. We will lead these DNA vaccines to the clinical studies through the process of safety confirmation in non-clinical studies.
References
・Uenaka, A., Wada, H., Isobe, M., Saika, T., Tsuji, K., Sato, E., Sato, S., Noguchi, Y., Kawabata, R., Yasuda, T., Doki, Y., Kumon, H,. Iwatsuki, K., Shiku, H., Monden, M., Jungbluth, A., Ritter, G., Murphy, R., Hoffman, E., Old, LJ. and Nakayama, E. : T cell immunomonitoring and tumor responses in patients immunized with a complex of cholesterol-bearing hydrophobized pullulan (CHP) and NY-ESO-1 protein. Cancer Immunity Vol. 7, p9, April 19, 2007
・Kitano, S., Kageyama, S., Nagata, Y., Miyahara, Y., Hiasa, A., Naota, H., Okumura, S., Imai, H., Shiraishi, T., Masuya, M., Nishikawa, M., Sunamoto, J., Akiyoshi, K., Kanematsu, T., Scott, AM., Murphy, R., Hoffman, EW., Old, LJ.,and Shiku, H. : HER2-Specific T-Cell Immune Responses in Patients Vaccinated with Truncated HER2 Protein Complexed with Nanogels of Cholesteryl Pullulan. Clin. Cancer Res. 2006; 12(24), 7397-7405, December 15, 2006
・Nishikawa, H., Kato, T., Takawa, I., Saito, K., Ikeda, H., Kuribayashi, K., Allen, P.M., Schreiber, R.D., Definition of target antigens for naturally occurring CD4+ CD25+ regulatory T cells. J.Exp.Med. 201(5)681-686, 2005.
・Nishikawa, H., Kato, T., Tawara, I., Ikeda, H., Kuribayashi, K., Allen, P.M., Schreiber, R.D., Old, L.J., and Shiku, H. : IFN-γcontrols the generation/activation of CD4+ CD25+ regulatory T cells in antitumor Immune response. J. Immunology 175, 4433-4440, 2005.
・Miyahara, Y., Naota, H., Wang, L., Hiasa, A., Goto, M., Watanabe, M., Kitano, S., Okumura, S., Takemitsu, T., Yuta, A., Majima, Y., Lemonnier, F. A., Boon and Shiku, H. : Determination of cellularly processed HLA-A2402-restricted novel CTL epitopes derived from two cancer germ lines, MAGE-A4 and SAGE. Clin. Cancer Res. 2005;11(15), 5581-5589, 2005.
・Nishikawa, H., Kato, T., Tawara, I., Takemitsu, T., Saito, K., Wang, L., Ikarashi, Y., Wakasugi, H., Nakayama, T., Taniguchi, M., Kuribayashi, K., Old, L.J., and Shiku, H. : Accelerated chemically induced tumor development mediated by CD4+ CD25+ regulatory T cells in wild-type hosts. Proc. Natl. Acad. Sci. USA. 102:9253-9257, 2005.
・Nishikawa, H., Kato, T., Tanida, K., Hiasa, A., Tawara, I., Ikeda, H., Ikarashi, Y., Wakasugi, H., Kronenberg, M., Nakayama, T., Taniguchi, M., Kuribayashi, K., Old, L.J., and Shiku, H. : CD4 +CD25 +T cells responding to serologically defined autoantigens suppress antitumor immune response. Proc. Natl. Acad. Sci. USA. 100:10902-10906, 2003.
・Wang L, Miyahara Y, Kato T, Wang L, Aota T, Kuribayashi K, Shiku H., Essential roles of tumor-derived helper T cell epitopes for an effective peptide-based tumor vaccine. Cancer Immun. Nov 21;3:16,2003
・Shiku, H., Importance of CD4+ helper T-cells in antitumor immunity. Int J Hematol. 77:435-8, 2003.
・Ishihara, M., Tawara, I., Wang, L., Takahashi, Y., and Shiku, H. : Elimination of CD4+ T cells may overcome suppression of anti-HER2 immune responses in tumor-bearing hosts. International J. Oncology 22:1135-1139, 2003.
・Ikuta,Y., Katayama, N.,Wang,L., Okugawa,T., Takahashi,Y., Schmitt,M., Gu,X., Watanabe,M., Akiyoshi,K., Nakamura,H., Kuribayashi,K., Sunamoto,J. and Shiku,H. :Presentation of a major histocompatibility complex class 1-binding peptide by monocyte-derived dendritic cells incorporating hydrophobized polysaccharide-truncated HER2 protein complex: implications for a polyvalent immuno-cell therapy. Blood 99:3717-3724, 2002.
・Mukai, K., Yasutomi, Y., Watanabe, M., Kenjo, A., Aota, T., Wang, L., Nishikawa, H., Ishihara, M., Fujita, T., Kuribayashi, K. and Shiku, H .: HER2 peptide-specific CD8(+) T cells are proportionally detectable long after multiple DNA vaccinations. Gene Ther. 9:879-888, 2002.
・Nishikawa, H., Tanida, K., Ikeda, H., Sakakura, M., Miyahara, Y., Aota, T., Mukai, K., Watanabe, M., Kuribayashi, K., Old, LJ. and Shiku, H. : Role of SEREX-defined immunogenic wild-type cellular molecules in the development of tumor specific immunity. Proc. Natl. Acad. Sci. USA. 98:14571-14576, 2001.
・Shiku, H., Wang, L., Ikuta, Y., Okuguwa, T., Schmidt, M., Gu, X., Akiyoshi, K., Sunamoto, J., Nakamura, H. :Development of a cancer vaccine: peptides, proteins, and DNA. Cancer Chemother. Pharmacol. 46(Suppl) S77-S82, 2000.
・Ikuta Y, Okugawa T, Furugen R, Nagata Y, Takahashi Y, Wang L, Ikeda H, Watanabe M, Imai S, Shiku H., A HER2/NEU-derived peptide, a K(d)-restricted murine tumor rejection antigen, induces HER2-specific HLA-A2402-restricted CD8(+) cytotoxic T lymphocytes. Int J Cancer. 15;87(4):553-8,2000
・Okugawa, T., Ikuta, Y., Takahashi, Y., Obata, H., Tanida, K., Watanabe, M., Imai, S., Furugen, R., Nagata, Y., Toyoda, N. and Shiku, H. : A novel human HER2-derived peptide homologous to the mouse K(d) -restricted tumor rejection antigen can induce HLA-A24 restricted cytotoxic T lymphocytes in ovarian cancer patients and healthy individuals. Eur. J. Immunol. 30 : 3338-3346, 2000.
・Gu, X., Schmidt, M., Hiasa, A., Nagata, Y., Ikeda, H., Sasaki, Y., Akiyoshi, K., Sunamoto, J., Nakamura, H., Kuribayashi, K. and Shiku, H. : A novel hydrophobized polysaccharide/oncoprotein complex vaccine Induces in vitro and in vivo cellular and humoral Immune responses against HER2-expressing murine sarcomas. Cancer Res. 58: 3385-3390, 1998.
・Nagata, Y., Furugen, R., Hiasa , A., Ikeda, H., Ohta , N., Furukawa, K., Nakamura, H., Furukawa, K., Kanematsu, T. and Shiku, H. : Peptides derived from a wild type murine proto- oncogene c-erB-2/HER2/neu can induce CTL and tumor suppression in syngeneic hosts. J. Immunol . 159: 1336-1343, 1997.
・Ikeda, H., Ohta, N., Furukawa, K., Miyazaki, H., Wang, L., Kuribayashi, K., Old, L.J. and Shiku, H. : Mutated mitogen-activated protein kinase: a tumor rejection antigen of mouse sarcoma. Proc. Natl. Acad. Sci. USA 94: 6375-6379, 1997.